India Orders Drugmakers to Meet Global Manufacturing Standards by January After Cough Syrup Deaths
The Central Drugs Standard Control Organisation (CDSCO) has instructed state regulators to inspect all drug manufacturing facilities and ensure adherence to the upgraded WHO-aligned standards. Companies that fail to comply will be ordered to suspend production until they meet the prescribed quality norms.
- By Newsdesk
- New Delhi
- Nov 11, 2025
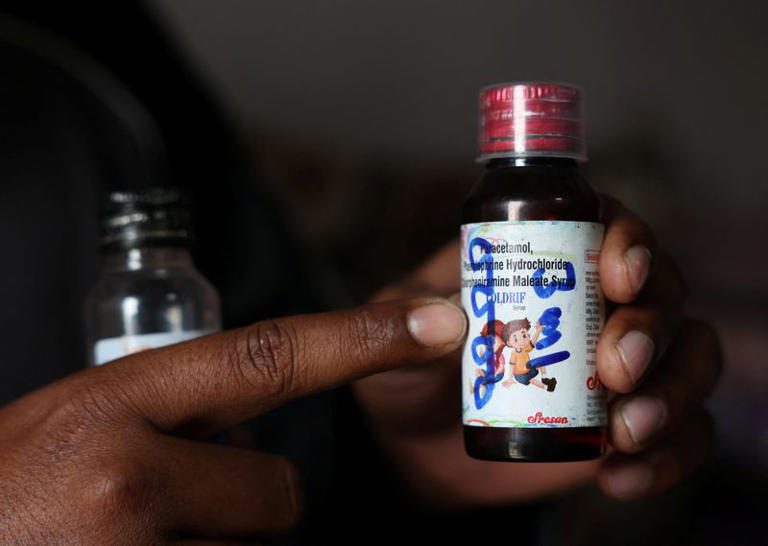
The Indian government has set a firm deadline requiring all pharmaceutical manufacturers in the country to comply with international Good Manufacturing Practices (GMP) by January 1, 2026, following a series of global incidents involving contaminated cough syrups linked to child deaths.
The Central Drugs Standard Control Organisation (CDSCO) has instructed state regulators to inspect all drug manufacturing facilities and ensure adherence to the upgraded WHO-aligned standards. Companies that fail to comply will be ordered to suspend production until they meet the prescribed quality norms.
The directive comes after several tragic incidents in which tainted cough syrups produced in India were found responsible for child fatalities overseas and domestically, raising concerns about lapses in quality control and regulatory oversight.
Under the new framework, large pharmaceutical manufacturers—those with annual turnover above ₹250 crore—were required to align with the standards by June 2024. Smaller units, which make up a significant portion of India’s drug industry, were granted an extension but must now comply by the January 2026 deadline, with no further leniency.
While the initiative aims to restore global confidence in Indian pharmaceuticals, smaller firms have expressed concerns about the high cost of compliance and the potential risk of shutdowns. Industry groups warn that enforcing the mandate without financial support could lead to job losses and higher drug prices.
The tightened rules will also require stricter documentation, improved testing of raw materials, and enhanced safeguards to prevent contamination. Recent inspections revealed significant lapses in quality management and process validation across several facilities.
India, which supplies more than one-fifth of the world’s generic medicines, is under growing international scrutiny. The government’s move signals a strong intent to protect public health, rebuild trust, and ensure that the “pharmacy of the world” meets global standards of safety and reliability.
